
New York faces another surge
Governor Hochul, Mayor Adams announce launch of new COVID-19 treatment hotline THE sixth wave of COVID-19 has started, with the highly contagious omicron variant – the BA.5 variant – now…
Covering the most noteworthy news stories in the health and wellness industry for the Filipino community.

Governor Hochul, Mayor Adams announce launch of new COVID-19 treatment hotline THE sixth wave of COVID-19 has started, with the highly contagious omicron variant – the BA.5 variant – now…

Whether it’s participating in a summer learning program, going to camp, or joining a new sports team, summer is an exciting time for children to learn, play, and grow through…
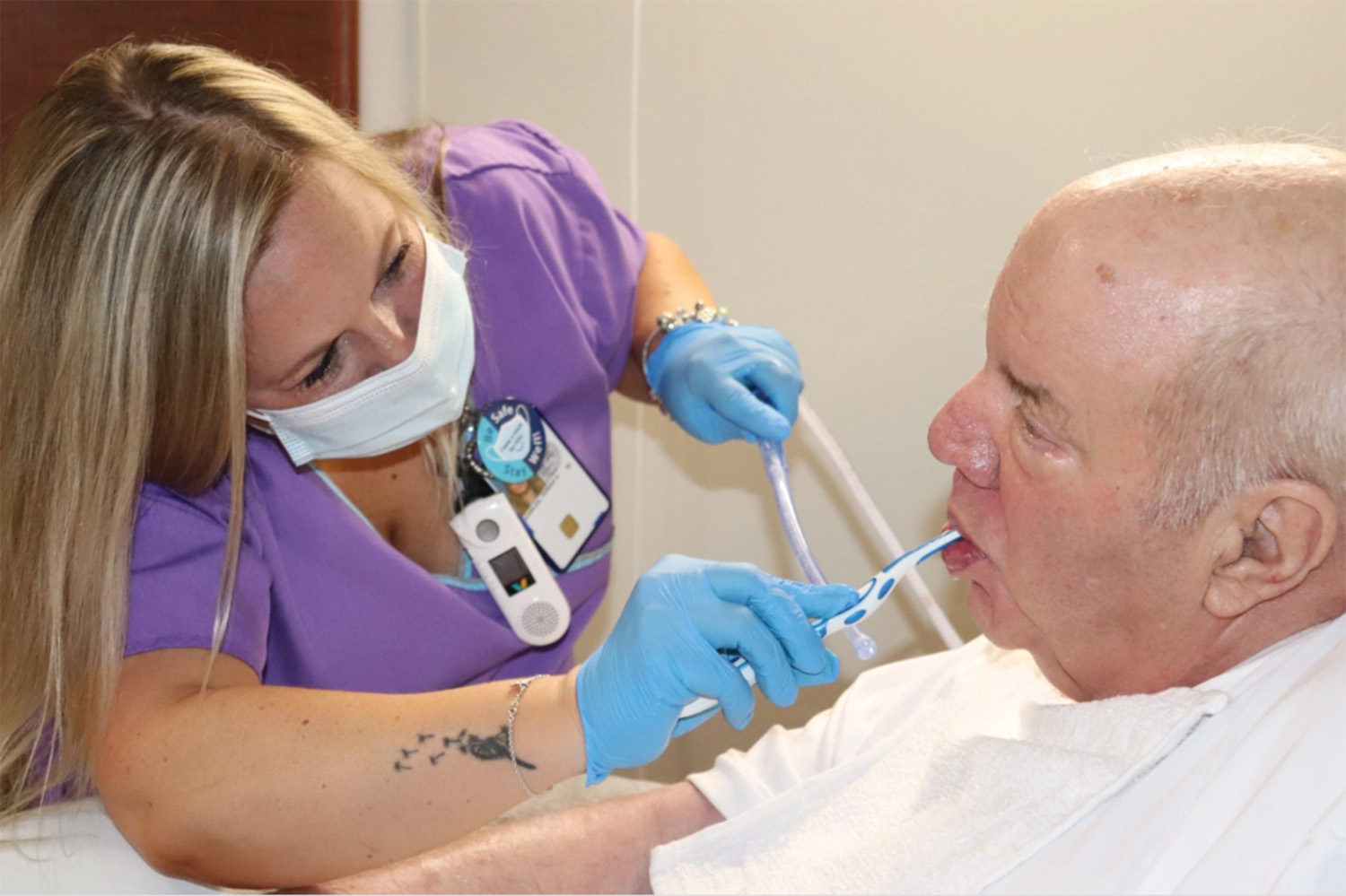
Four years ago, when Karen Giuliano went to a Boston hospital for hip replacement surgery, she was given a pale-pink bucket of toiletries issued to patients in many hospitals. Inside…

WITH summer in full swing, Los Angeles County officials are warning residents to continue to practice COVID-19 safety measures — or else the indoor mask-wearing mandate could be reinstated by…

By Mark Hedin/Ethnic Media Services “You might be done with COVID, but COVID is not done with you.” So said Denny Chan, directing attorney with Justice in Aging, at a press briefing co-hosted…
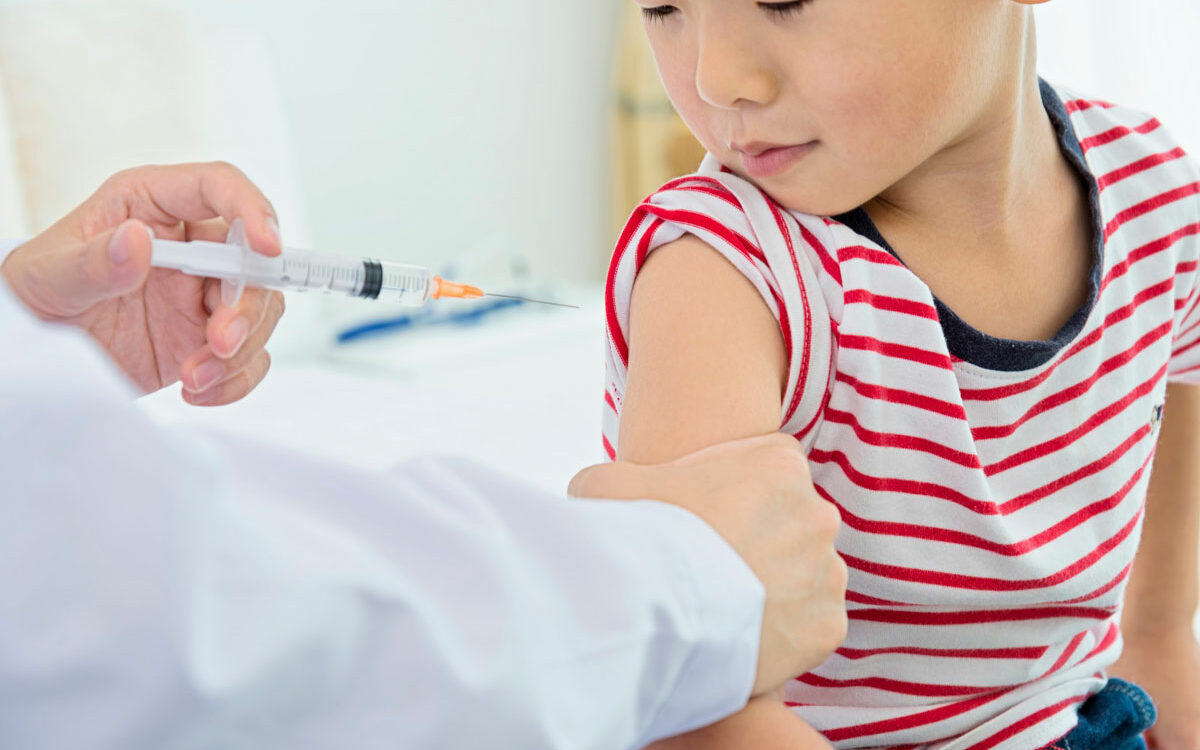
AFTER more than a year of clinical trials, children in the United States between the ages of 6 months and 4 years old are now permitted to receive the COVID…

By Judith Graham/ KHN ALMOST half of older adults — more than 26 million people 65 and older — have prediabetes, according to the Centers for Disease Control and Prevention….

CHILDREN under the age of 5 years old will now be able to receive the COVID-19 vaccine. This comes after an independent panel of advisers to the U.S. Centers for…

THE Southern Nevada Health District has begun rolling out COVID-19 vaccines to children under the age of 5, following authorization from health officials. Last week, an independent panel of advisers…

A FEW months ago, it seemed as though the country was poised to finally tame the pandemic, after two years of restrictions and tens of billions in government spending. The…

MORE than 800,000 COVID-19 cases have been reported in the San Diego region since the start of the pandemic, the County Health and Human Services Agency reported on Thursday, June…

9 INCREASES in new Omicron variants continue to fuel high transmission in Los Angeles County, with cases and hospitalizations currently much higher than they were at this time last year,…

With high rates of transmission across Los Angeles County, positive cases and outbreaks are increasing at skilled nursing facilities, the public health department reported on Friday, June 10. For the…
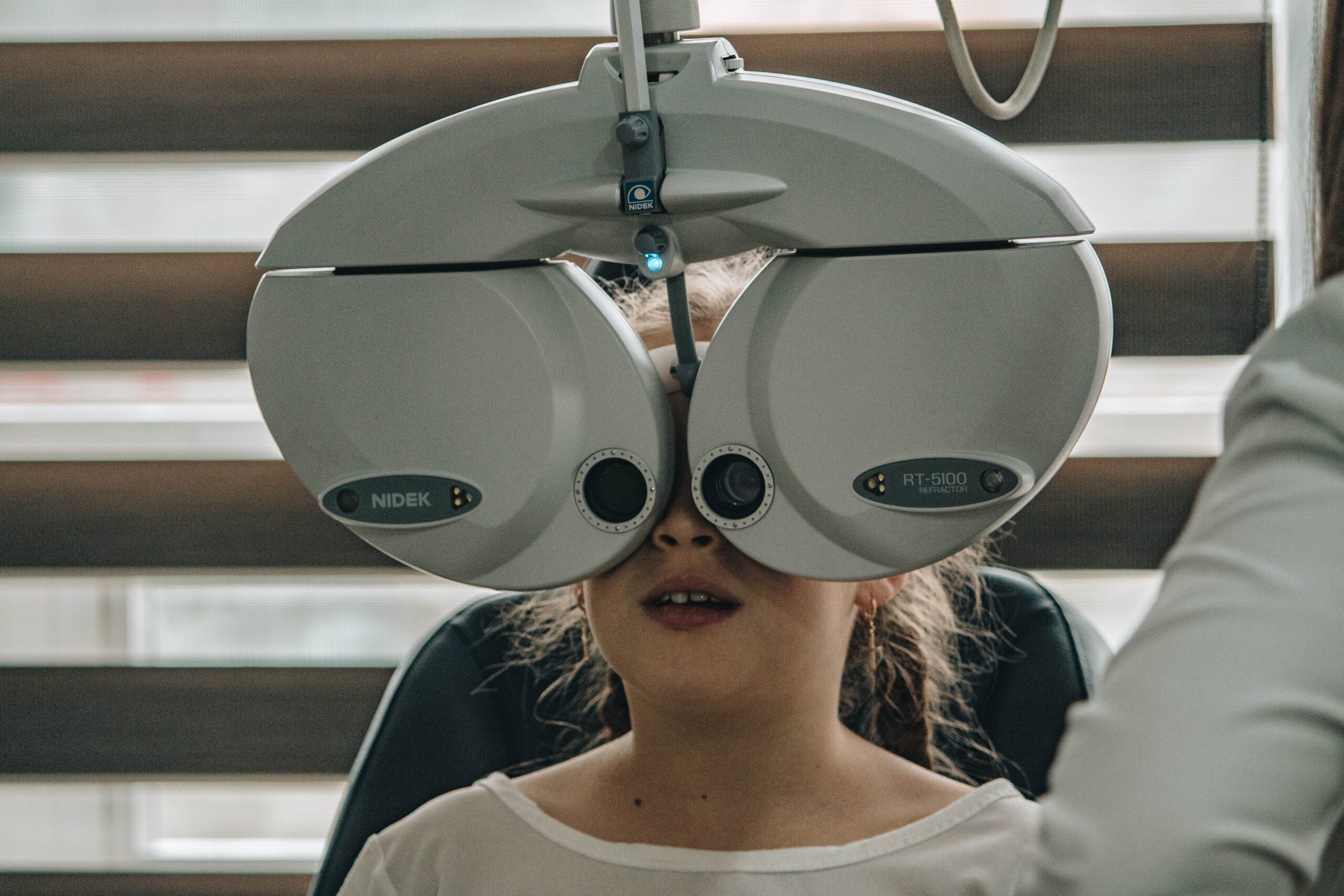
Jessica Oberoi, 13, can’t exactly remember when her eyesight started getting blurry. All she knows is that she had to squint to see the whiteboard at school. It wasn’t until…
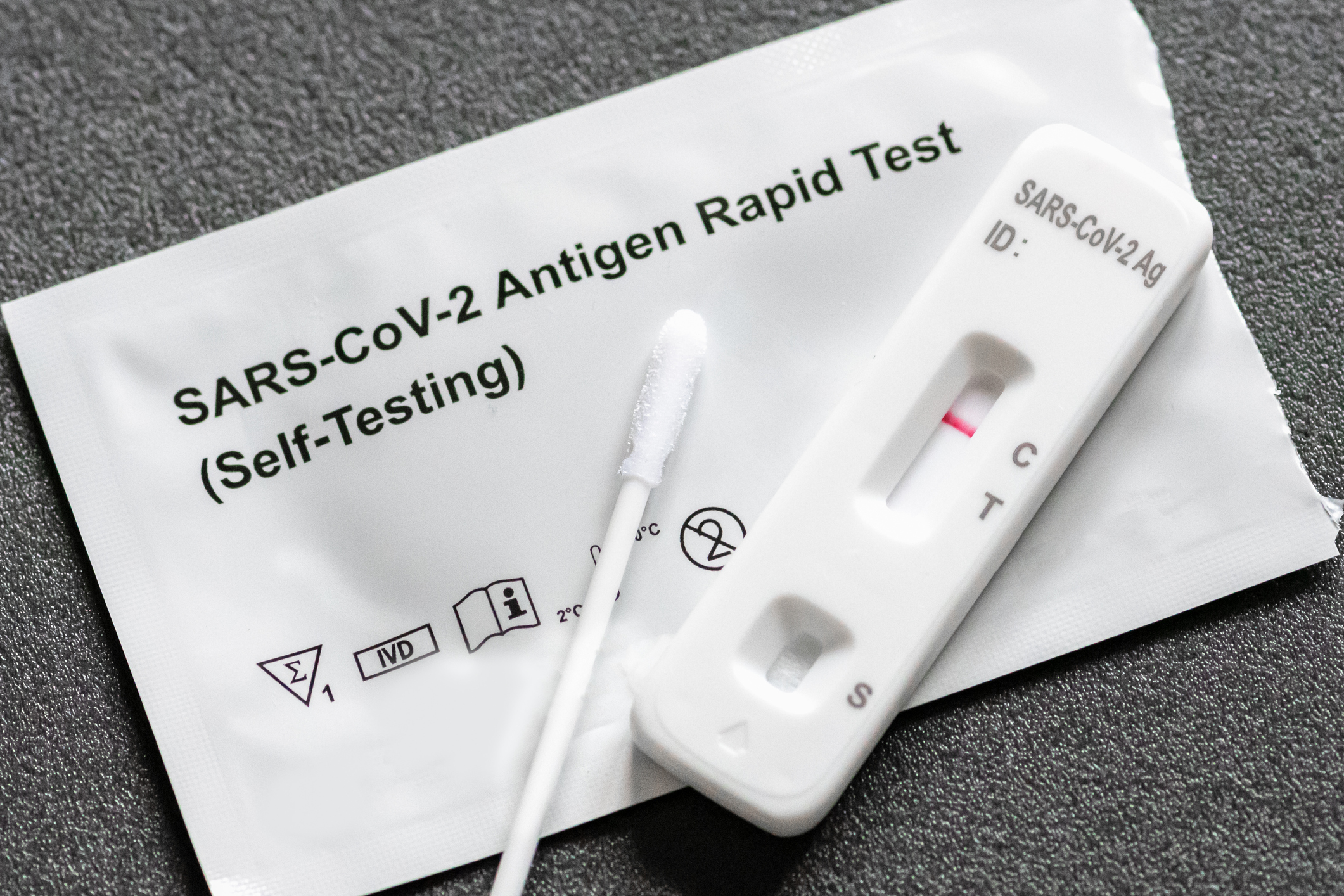
WITH summer approaching and the increase in public gatherings, Los Angeles County is reminding residents anew to get tested. At-home testing for residents that have an exposure to a known…
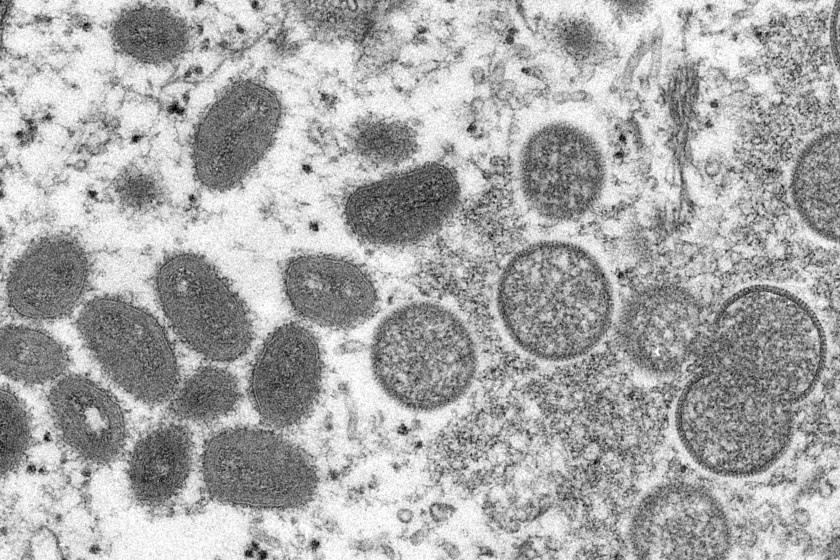
The Los Angeles County Department of Public Health has confirmed the first presumptive case of monkeypox infection in Los Angeles County and is awaiting final confirmation from the Centers for Disease Control…

Older adults who have survived COVID-19 are more likely than younger patients to have persistent symptoms such as fatigue, breathlessness, muscle aches, heart palpitations, headaches, joint pain, and difficulty with…

WITH cases and outbreaks remaining high and many opportunities over the Memorial Day Holiday to gather, Los Angeles County residents are being encouraged to layer in safety measures to prevent…
![[AD] Revolutionary cancer treatment, good news for hopeless patients](https://asianjournal.com/wp-content/uploads/2022/05/Dr.-Chang-seok-Kim.jpg)
EVERYONE suffers from large and small ailments during their lifetime. In particular, cancer has long been a terrifying disease that has long been thought to be an incurable disease, with…

AMONG other things, the month of May is an important month for two observances: celebrating Asian American heritage and spreading mental health awareness. In the United States, Asian Pacific American…

LOS ANGELES – California Health Collaborative Every Woman Counts Los Angeles has joined forces with Comprehensive Community Health Centers (CCHC) and Cedars Sinai Cancer to celebrate Women’s Health and Asian-American/Pacific…

THE New York City Commissioner of Health and Mental Hygiene on Monday, May 16 issued an advisory strongly recommending that all New Yorkers wear masks in all indoor public settings…
Even as top U.S. health officials say it’s time America learns to live with the coronavirus, a chorus of leading researchers say faulty messaging on booster shots has left millions of older…

Targeted outreach and consistent care will provide San Franciscans in need of behavioral health services with a far more robust and proactive support system so they can avoid a cycle…
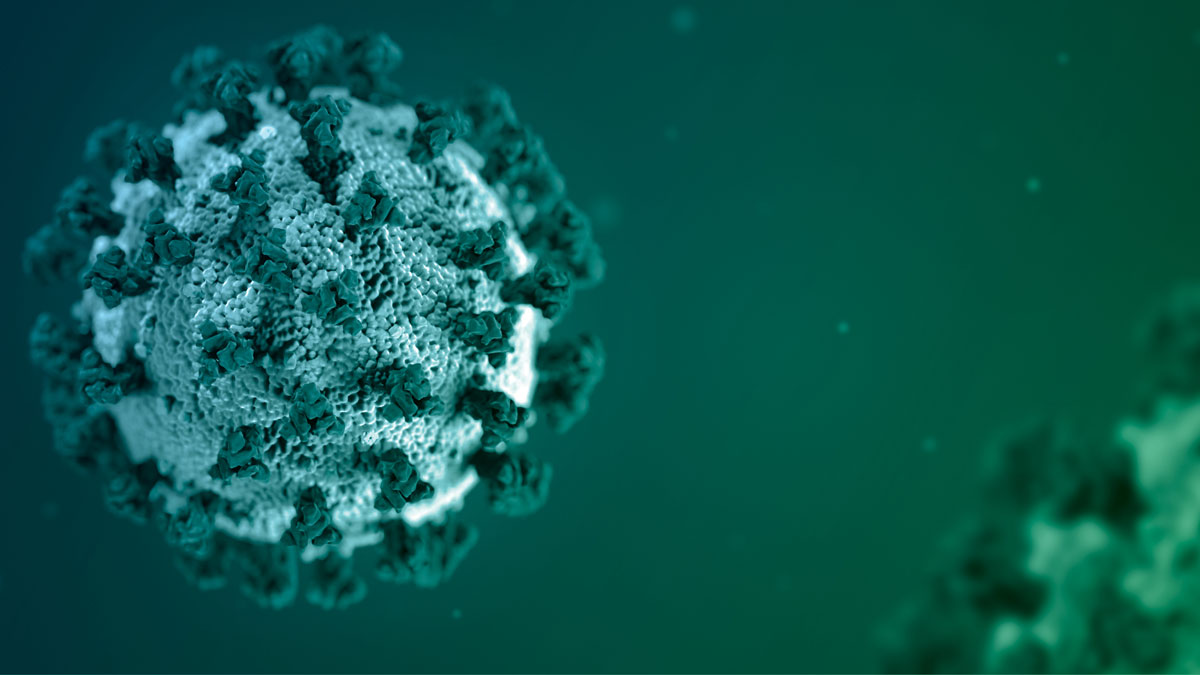
TWO years into the coronavirus pandemic, Americans can be forgiven if they’ve lost track of the latest variants circulating nationally and around the world. We’ve heard of the alpha, beta,…
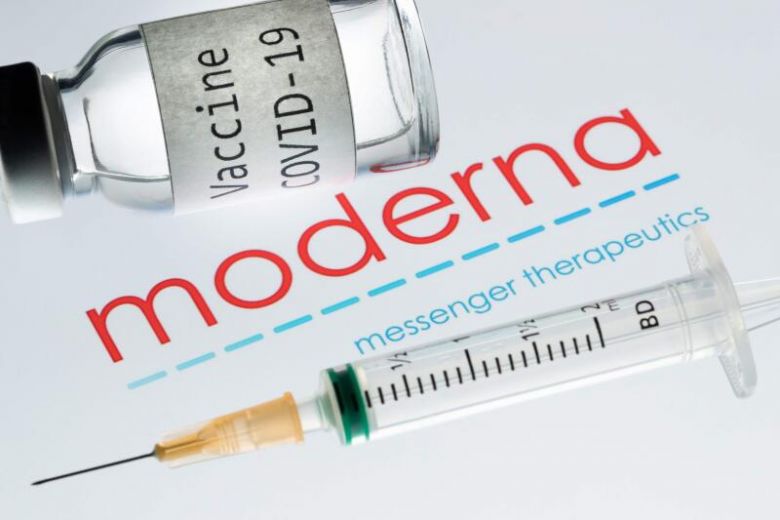
By Jenny Manrique/Ethnic Media Services THE medical community is celebrating drug maker Moderna’s recent announcement that it is seeking FDA approval for its COVID-19 vaccination for younger kids, though…

COVID-19 cases continue to rise in the United States with the Northeast states recording the highest seven-day case rates per 100,000. New York City is experiencing an uptick in positive…
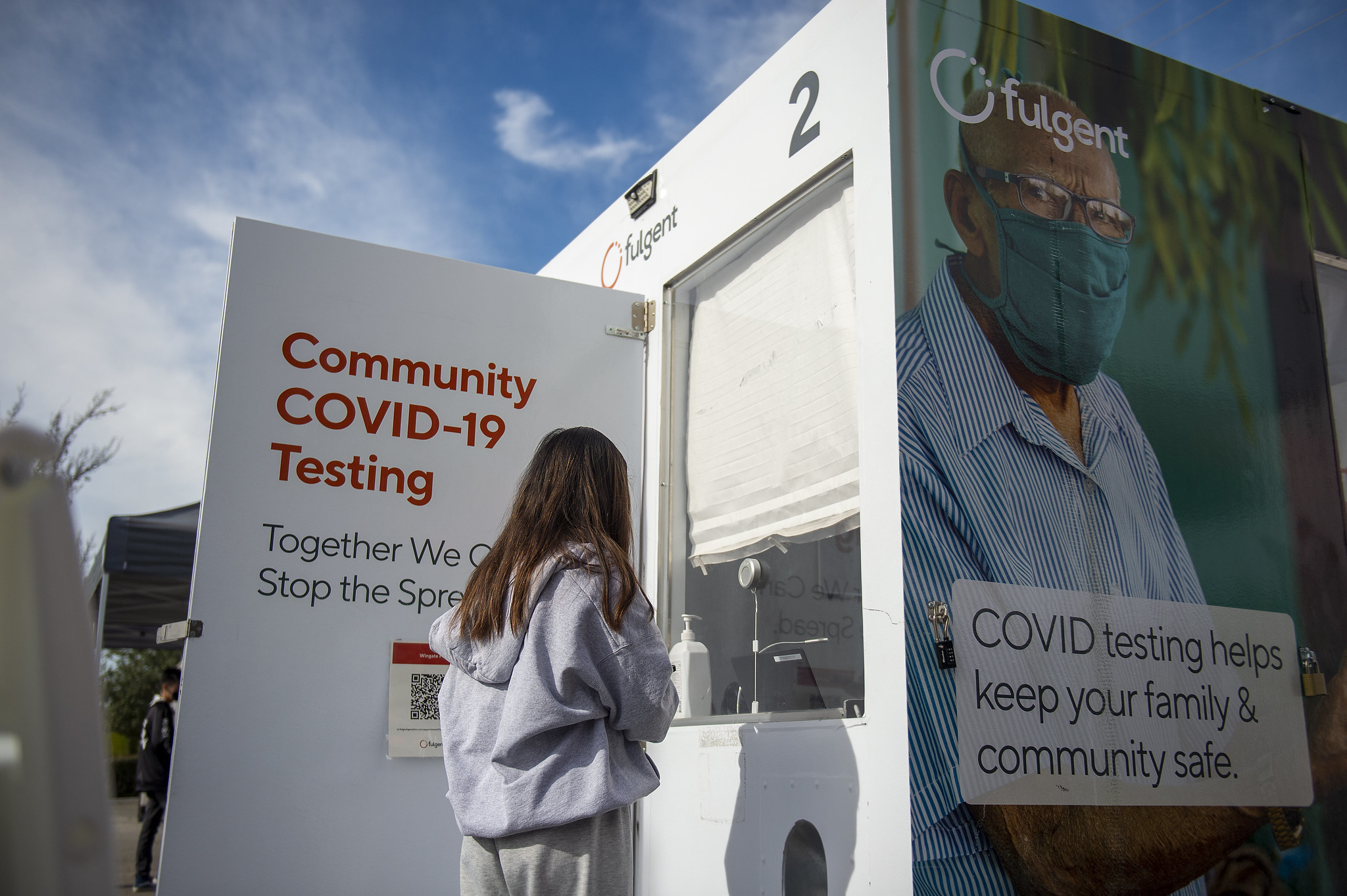
WITH the easing of COVID-19 restrictions, Los Angeles County is observing another increase of new cases and test positivity rates. As of Tuesday, April 12, the average number of daily…

By Bram Sable-Smith and Rachana Pradhan/Kaiser Health News Korra Elliott has tried to avoid seeing a doctor while waiting to get on Medicaid. She worries she can’t afford more bills without any insurance…
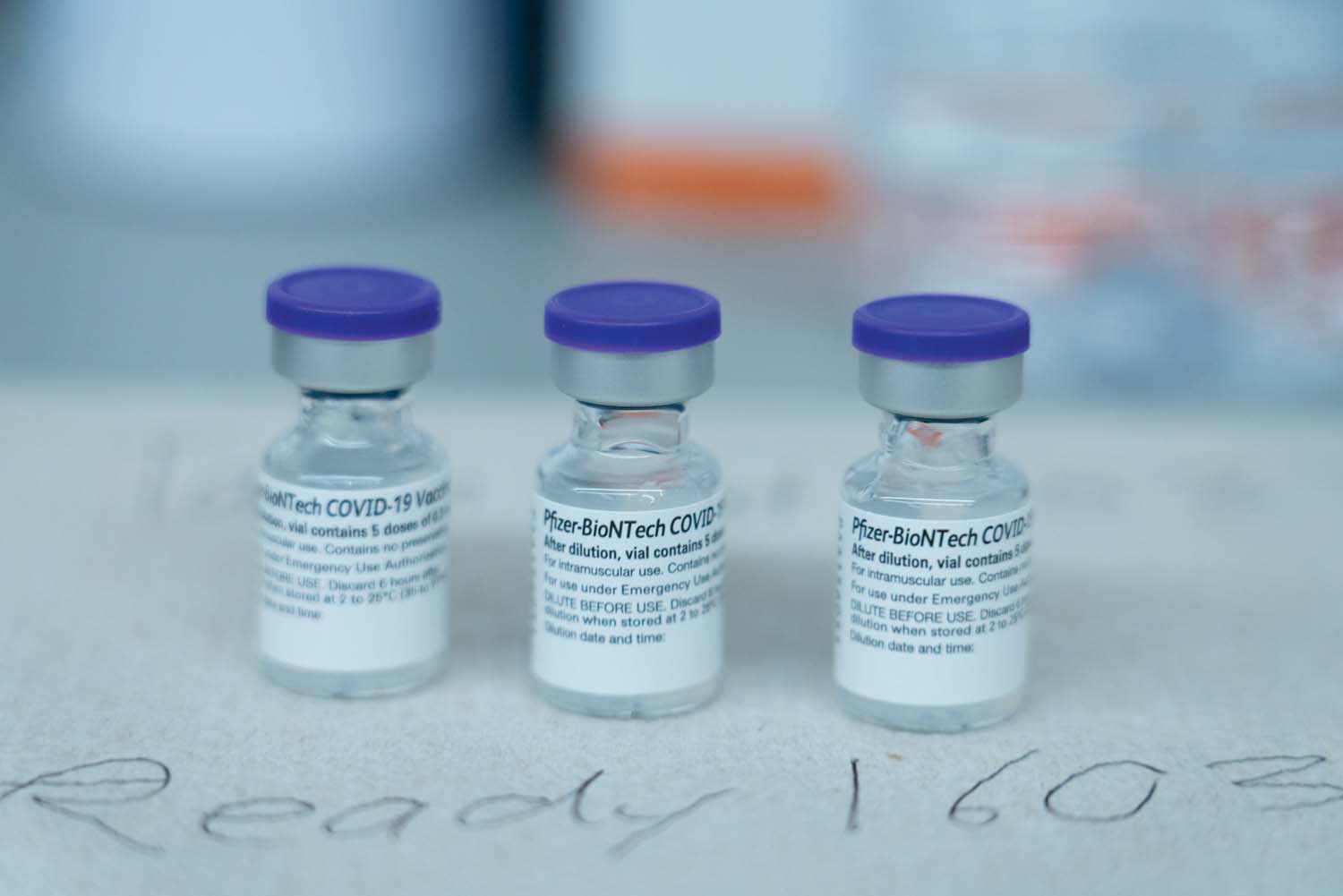
OLDER people may soon be able to get a fourth COVID booster after it was announced that Pfizer and its partner, BioNTech, plan to ask the United States Food and…